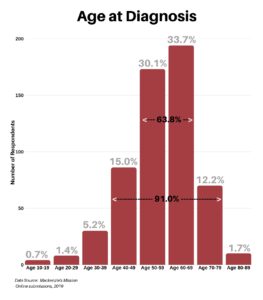
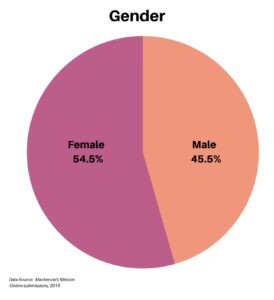
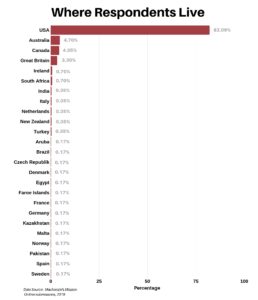
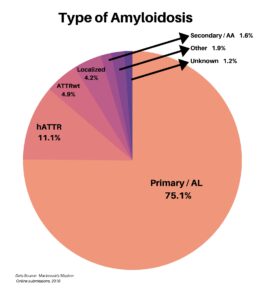
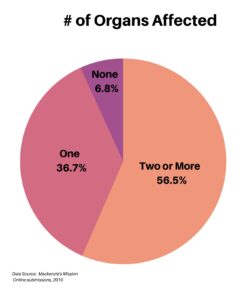
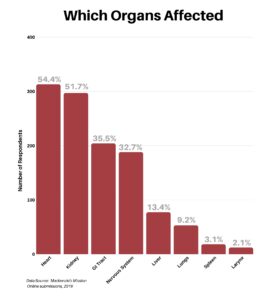
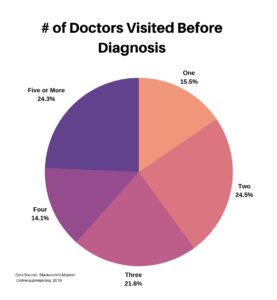
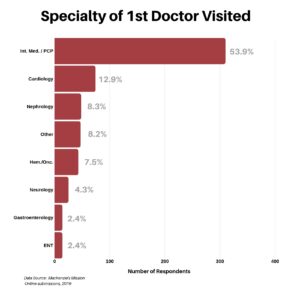
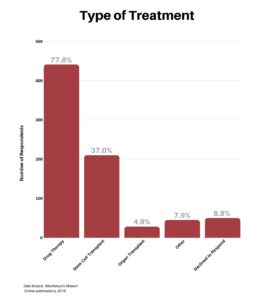
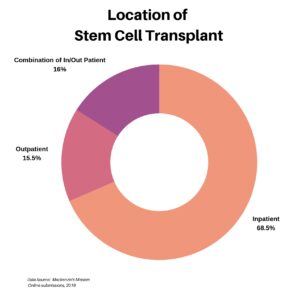
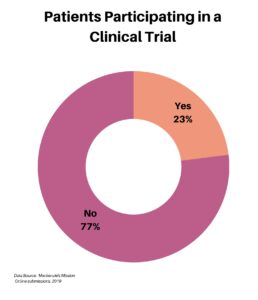
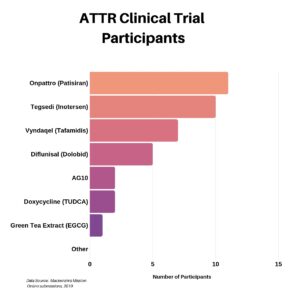
Clinical research is simply medical research involving people. There are two types, clinical studies (aka observational studies) and clinical trials. In this blog, we explore clinical trials and the basics of what you need to know.
WHAT ARE CLINICAL TRIALS?
According to the National Institutes of Health (NIH), clinical trials are research studies performed on people that are aimed at evaluating a medical, surgical, or behavioral intervention. Clinical trials are the primary way that researchers find out if a new treatment, like a new drug or medical device (e.g., a pacemaker) is safe and effective in people. Often a clinical trial is used to learn if a new treatment is more effective and/or has less harmful side effects than the standard treatment. Other clinical trials test ways to find a disease early, sometimes before there are symptoms. Still, others test ways to prevent a health problem before it begins. A clinical trial may also look at how to make life better for people living with a life-threatening disease or a chronic health problem.
WHY CLINICAL TRIALS ARE IMPORTANT
Clinical trials permit researchers to test the safety and effectiveness of new therapies. They also allow for a rigorous evaluation through patient participation. Bottom line: it is only after the extensive evaluation and testing from a clinical trial that the FDA will approve the widespread use of any new therapy.
According to Dr. Morie A. Gertz at the Mayo Clinic:
Advancing the medical care for all patients requires participation in clinical trials. Only through clinical trials can we further improve the available therapy options for current and all future patients. Clinical trials seek to answer questions about the natural history and biology of the disease as well as important questions regarding outcomes was all available new therapies. All current treatments received by our amyloid community were derived through others participation in clinical trials. Clinical trials are not only an opportunity to get the cutting edge therapy but is a way to “pay it forward“ for future generations of Patients.
WHAT ARE THE PHASES OF CLINICAL TRIALS?
All clinical trials must be approved by the U.S. Food and Drug Administration (FDA) before they can begin. Prior to that decision, scientists perform laboratory tests and studies in animals to test a potential therapy’s safety and efficacy. Assuming favorable outcomes, the FDA then gives approval for a clinical trial involving humans.
Clinical trials are comprised of four phases to test a treatment, find appropriate dosages, and detect side effects. If following the completion of the first three phases, researchers find the drug or intervention to be safe and effective, the FDA approves it for clinical use and continues to monitor its effects. Overall, the duration of a clinical trial spans years.
- A Phase I trial tests an experimental treatment on a small group of often healthy people (20 to 80) to judge its safety and side effects and to find the correct drug dosage.
- A Phase II trial uses more people (100 to 300). While the emphasis in Phase I is on safety, the emphasis in Phase II is on effectiveness. This phase aims to obtain preliminary data on whether the drug works in people who have a certain disease or condition. These trials also continue to study safety, including short-term side effects. This phase can last several years.
- A Phase III trial gathers more information about safety and effectiveness, studying different populations and different dosages, using the drug in combination with other drugs. The number of subjects usually ranges from several hundred to about 3,000 people. If the FDA agrees that the trial results are positive, it will approve the experimental drug or device.
- A Phase IV trial for drugs or devices takes place after the FDA approves their use. A device or drug’s effectiveness and safety are monitored in large, diverse populations. Sometimes, the side effects of a drug may not become clear until more people have taken it over a longer period of time.
WHY PEOPLE CHOOSE TO JOIN A CLINICAL TRIAL
There are many reasons why people choose to join a clinical trial. Some join a trial because the treatments they have tried for their health problem did not work. Others participate because there is no treatment for their health problem. Some studies are designed for, or include, people who are healthy but want to help find ways to prevent a disease that may be common in their family. By being part of a clinical trial, participants may access new treatments before they are widely available. Especially with a rare disease such as amyloidosis, clinical trials may offer a meaningful impact to a patient’s quality of life.
In addition, people may feel that participating in a clinical trial allows them to play a more active role in their own health care. Participants may receive more frequent health check-ups and closer monitoring through the clinical trial. Other people say they want to help researchers learn more about certain health problems. Whatever the motivation, when choosing to participate in a clinical trial, one becomes a partner in scientific discovery. This can also help future generations lead healthier lives. Major medical breakthroughs could not happen without the generosity of clinical trial participants—young and old.
In the words of Dr. Vaishali Sanchorawala at The Amyloidosis Center at Boston University School of Medicine and Boston Medical Center:
Clinical trials allow researchers and physicians to test the safety and effectiveness of new, promising drugs. Before any drug can be approved, it must be rigorously tested in clinical trials. Without the participation of patients, new treatments and cures will never happen. In addition, participating in a clinical trial may be a great way for patients to access new treatments before they become available. Especially in a rare disease such as amyloidosis, clinical trials can be a vital resource for the care of patients.
POTENTIAL RISKS OF A CLINICAL TRIAL
There are no guarantees of success from a clinical trial, and there are risks. For starters, there may be serious side effects. Also, the therapy may not improve upon current treatment, or may not even work at all. Finally, as a clinical trial participant, you may be part of the control group, which means either standard treatment or no-treatment placebo. In other words, there are no assurances you would receive the new therapy.
FINDING A CLINICAL TRIAL
Thanks to the internet, folks can find lots of information regarding the wide array of open clinical trials. So much so that it may be overwhelming. Particularly with regards to amyloidosis, casting such a wide net may not be the most productive approach. Since finding an appropriate clinical trial is not as easy for rare diseases such as amyloidosis, here are a few excellent places to start.
- My Amyloidosis Pathfinder (MAP). Developed by the Amyloidosis Research Consortium (ARC), MAP helps patients discover and learn about amyloidosis-related clinical trials. After answering a short questionnaire, MAP matches patients to trials specific to their condition and ones for which they may be eligible.
- Boston University / Boston Medical Center. A recognized Center of Excellence for amyloidosis, BU has an ongoing robust array of clinical trials for different types of amyloidosis.
- Mayo Clinic. A recognized Center of Excellence for amyloidosis, Mayo Clinic has an extensive clinical trial program in the area of amyloidosis.
- ClinicalTrials.gov. This resource, provided by the U.S. National Library of Medicine, is a database of over 250,000 privately and publicly funded clinical studies conducted around the world (in all 50 states in the U.S. and 204 countries).
INFORMED CONSENT PROCESS
The informed consent process is a key part of the safeguard of a clinical trial. Before joining a clinical trial, each participant will be told what to expect (e.g., treatments, tests) and what might happen (e.g., benefits and risks, including possible side effects). It is also the point where participants should ask ample questions about the trial, which the clinical trial coordinator should be more than willing to answer.
Below is a list of questions compiled from sources, including the NIH and The Clinical Study Center, recommending what patients should consider asking before consenting to participation in a clinical trial.
- What is the purpose of the study?
- Who is sponsoring the study, and who has reviewed and approved it?
- Who will be in charge of my care?
- What treatment or tests will I have? Will they hurt?
- What are the chances I will get the experimental treatment?
- What are the possible risks, side effects, and benefits of the study treatment compared to my current treatment?
- How will I know if the treatment is working?
- How will you protect my health while I am in the study?
- What happens if my health problem gets worse during the study?
- How will the study affect my everyday life?
- How long will the clinical trial last?
- What will happen after the conclusion of the study?
- Where will the study take place? Will I have to stay in the hospital?
- Will you provide a way for me to get to the study site if I need it?
- Will being in the study cost me anything (e.g., treatment, tests, travel)? If so, will I be reimbursed for all expenses (including other charges such as child care)? Will my insurance cover my costs?
- Can I take my regular medicines while in the trial?
- Who will be in charge of my care while I am in the study? Will I be able to see my own doctor?
- Will you follow up on my health after the end of the study?
- Will you tell me the results of the study?
- Whom do I call if I have more questions?
- How will you keep my doctor informed about my participation in the trial?
- Does the study compare standard and experimental treatments?
- If I withdraw, will this affect my normal care?
- What are the chances that I will receive a placebo?
- What steps ensure my privacy?
Taking part in a clinical trial is solely the decision of the participant, although they may want to discuss it with their medical team prior to finalizing a decision. If one decides to join the trial, they will be required to sign an informed consent form that presents the key facts of the study and indicates they have been told all of the details and want to be part of the study. Importantly, the informed consent form is NOT a contract. Participants can leave the trial at any time and for any reason without being judged or put in a difficult position regarding medical care. Researchers much keep health and personal information private. Also, during the trial, participants have the right to learn about new risks or findings that emerge.If researchers learn that a treatment harms any of the participants, they’ll be removed from the study.
PARTICIPANT PROTECTION & SAFETY
Before committing to participate in a clinical trial, it is important to understand participant safety. Congress has put laws in place to protect against abuses, and today every clinical investigator is required to monitor and make sure that every participant is safe. These safeguards are enforced by the Federal Government. Every clinical trial follows a protocol that describes what the researchers will do. The principal investigator, or head researcher, is responsible for making sure that the protocol is followed.
In addition, there are multiple scientific oversight groups to aid in the control of clinical trials.
- Institutional Review Board (IRB): Comprised of doctors, scientists, statisticians, and lay people, IRBs provide scientific oversight for all clinical trials in the United States. IRB members regularly review studies and their results, making sure risks (or potential harm) are minimized.
- Office for Human Research Protections (OHRP): The U.S. Department of Health and Human Services’ (HHS) Office for Human Research Protections (OHRP) oversees all research done or supported by HHS. The OHRP helps protect the rights, welfare, and well-being of research participants. They provide guidance and oversight to the IRBs, develop educational programs and materials, and offer advice on research-related issues.
- Data Safety Monitoring Board (DSMB): Comprised of research and study topic experts, this board is required for every NIH phase III clinical trial. Their role is to review data from a clinical trial for safety problems or differences in results among different groups of relevant studies. If they find that the experimental therapy is not working or is harming participants, they will halt the trial right away.
- Food and Drug Administration (FDA): In the United States, the FDA provides oversight for clinical trials that are testing new medicines or medical devices. They review applications before any testing on humans is done, checking to ensure a proposed clinical trial has proper informed consent (see earlier) and protection for human subjects. In addition, the FDA provides oversight and guidance at various stages throughout the trial.
Scientific oversight informs decisions about a trial while it’s underway. For example, some trials are stopped early if benefits from a strategy or treatment are obvious. That way, wider access to the new strategy can occur sooner. Sponsors also may stop a trial, or part of a trial, early if the strategy or treatment is having harmful effects. Protecting the safety of people who take part in clinical trials is a high priority for all involved. Each trial has scientific oversight, and patients also have rights that help protect them.
DECIDING WHO PARTICIPATES IN CLINICAL TRIALS
After signing the informed consent form, the clinical staff will screen the candidate against the clinical trial criteria. The screening may involve cognitive and physical tests. Inclusion criteria for a trial might include age, stage of disease, gender, genetic profile, family history, and whether or not the candidate has a study partner who can accompany them to future visits. Exclusion criteria might include factors such as specific health conditions or medications that could interfere with the treatment being tested. Generally, individuals can participate in only one trial or study at a time. Different trials have different criteria, so being excluded from one trial does not necessarily mean exclusion from another.
Clinical trials need numbers … many volunteers must be screened to find enough people for a study, and with rare diseases such as amyloidosis, important trials are often significantly delayed due to a lack of participants. This can seriously slow down the rate at which new drugs are discovered, tested, and made available to patients.
CONCLUSION
Not all clinical trials have successful outcomes. However, every disease-related drug and therapy treatment prescribed today is the result of clinical research. Clinical trials are absolutely necessary to determine that a treatment is safe and that it has a real positive effect on a particular disease, better than that observed by a placebo or the current standard of care.
Final thoughts from Isabelle Lousada, founder and CEO of Amyloidosis Research Consortium:
Clinical trials play a critical role in evaluating novel therapies, establishing the best treatment pathways, and increasing our knowledge about amyloidosis.
SOURCES
Amyloidosis Research Consortium
Boston University / Boston Medical Center, Amyloidosis Center
The Clinical Study Center
Mayo Clinic
National Institute on Aging
National Institutes of Health
U.S. National Library of Medicine